| 2. Understanding Corrosion | |
|
2.5.3 Electric Double Layer |
|
Helmholtz Double Layer
When a metal dissolves continuously, it may become increasingly negatively charged because of passage of continuous positive ions in the solution. The excess negative charge on the metal surface balances the excess positive charge and the solution side of the interface and hence, a separation of charges exists.
The electrons orient themselves opposite a layer of cations of equal and opposite charges on the solution side of the interface. The electric field which is created consists of two layers of charges; hence it has been given a name double layer. The separation of charges in the double layer can be compared to parallel plates in a capacitor as it was done by Helmholtz. The double layer is also called Helmholtz Double Layer.
The current would flow during charging, when a potential is applied at the electrode solution interface. There would be a a charge qm on the metal electrode and qs on the solution so that
qm = - qs
The charge represents either an excess or deficiency of electons and it is located in a very thin layer of metal surface (< 0.1 Ao). The charge is made up of either excess cations or anions at the metal/electrode interface.
Water Molecules
It is known that water is a dipolar molecule. The oxygen-end of water molecule forms the negatively charged end and the hydrogen-end the positively charged end. Due to the dipole nature, the water molecule is attracted towards the electrode and contributes to the potential difference across the double layer.
The orientation of the water molecule depends on the charge on the metal surface. If the metal has large negative charge, the H2O molecules would be oriented with the positive ends (hydrogen) towards the metal and negative ends (oxygen) towards the large positive charge.
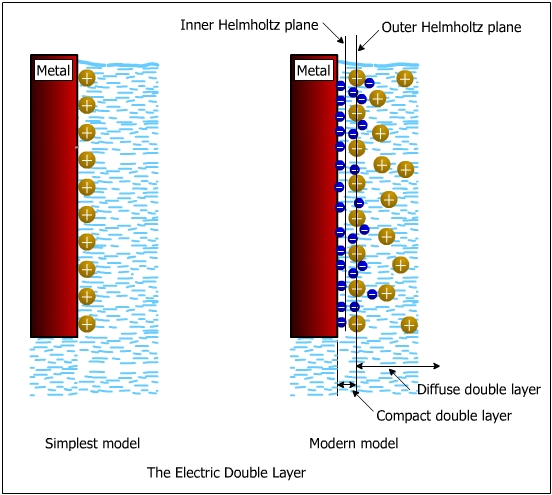
The water molecules are attracted towards the metal electrode and contribute to the potential difference. The water molecules form the first adsorbed layer on the metal surface. The cations which are hydrated and attracted towards the metal surface are limited in their approach to the metal surface because of the presence of water molecules on the metal surface. When electrostatic interaction operates, ions from the solution phase approach the electrode only as far as their salvation sheath allow. The surface arrays of these ions are thus cushioned from the electrode surface by a layer of solvent molecules. The line drawn through the center of such solvated cations at distance of closest approach is called “outer Helmholtz plane.”
The anions are specifically adsorbed sometimes in the water molecules (solvent). The water molecules sometimes contain the specifically adsorbed anions. The water molecules form the inner Helmholtz layer. The line drawn through the center of these molecules is called the “inner Helmholtz plane.” The outer Helmholtz plane φ2 (OHP) represents the locus of the electrical centers of the positive charges. This plane resides at a fixed distance from the metal due to water molecules that are between the surface of the metal and ions. The outer Helmholtz plane (OHP) is significantly affected by hydrated cations M z+ (hydrated).
To summarize, the double layer consists of three constituents:
a) A inner layer (inner Helmholtz layer) in which the potential changes linearly with the distance. It comprises the absorbed Water molecules and sometimes the specifically adsorbed anions.
b) An outer Helmholtz layer. It comprises hydrated (solvated) cations. The potential varies linearly with the distance.
c) An outer diffuse layer, also called the Guoy-Chapman layer, which contains excess cations or anions distributed in a diffuse layer. The potential varies exponentially with the distance, F.
|
|