| 5. Cathodic Protection | |
|
5.1 Introduction [1/3] |
|
Cathodic protection is a proven method of corrosion control and widely practiced with successful protection of oil and gas pipelines and underground metallic structures.
Definition
#1: Cathodic protection may be defined as the protection of metal surface by making it completely cathodic and eliminating all anodic areas.
#2: Cathodic protection may also be defined as the transfer of corrosion from the structure to be protected to another structure.
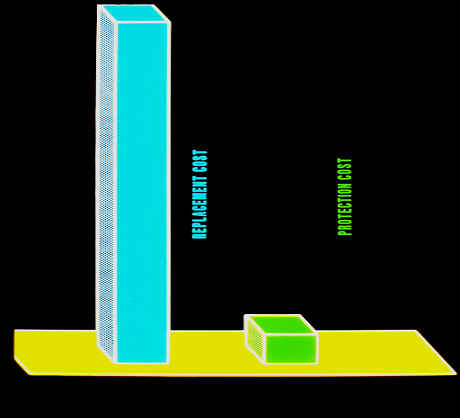
Following figure gives an indication of the cost of cathodic protection as compared to the replacement cost.

Why underground structures corrode?
Consider a section of a buried pipeline as shown below.
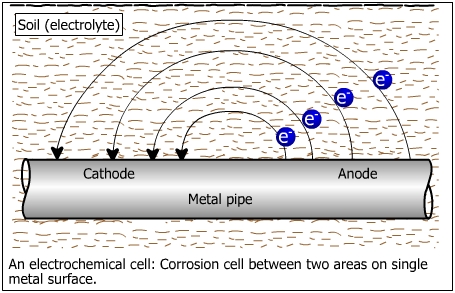
The pipeline represents a corrosion cell combining an anode, a cathode, a metallic path and electrolyte. Compare the above figure with that of a simple electrochemical cell shown below.
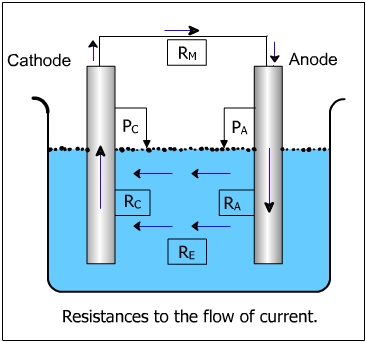
The corrosion cells are batteries in principle, the essential action being the electron transfer between the parts of the corrosion cell; the total number of electrons remaining constant. The driving force is the potential difference in the anodic and cathodic areas of the pipe. The total resistance of the corrosion cell is comprised of the resistance of the electrolyte and the metallic path (Rtotal = Re + Rm).
|
|